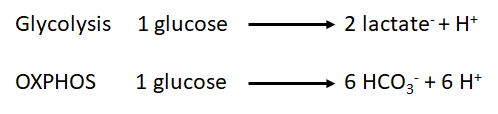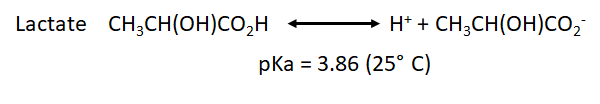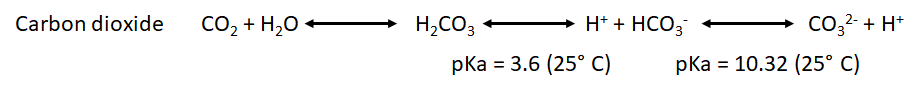Description
Volume-specific proton flux is measured in a closed system as the time derivative of proton concentration, expressed in units [pmol·s-1·mL-1]. Proton flux can be measured in an open system at steady state, when any acidification of the medium is compensated by external supply of an equivalent amount of base. The extracellular acidification rate (ECAR) is the change of pH in the incubation medium over time, which is zero at steady state. Volume-specific proton flux is comparable to volume-specific oxygen flux [pmol·s-1·mL-1], which is the (negative) time derivative of oxygen concentration measured in a closed system, corrected for instrumental and chemical background.
pH is the negative logarithm of proton activity. Therefore, ECAR is of interest in relation to acidification issues in the incubation buffer or culture medium. The physiologically relevant metabolic proton flux, however, must not be confused with ECAR.
Abbreviation: JH+
Reference: Gnaiger 2014 MitoPathways
Proton flux versus glycolytic flux
- Measured changes in pH over time (ECAR) must be transformed from the logarithmic scale to the linear scale of proton flux.
- Measurement of extracellular proton flux and glycolytic flux are related under specifically controlled conditions. Such conditions must be carefully evaluated, may require modifications of protocols, and must be corrected for acid-base reactions unrelated to glycolytic flux.
- The measurement of proton flux alone is not sufficient to determine if the origin of the protons is the glycolysis or other sources. For example, the carbon dioxide formed during the mitochondrial respiration acts as a net donor of protons into the media and as consequence has to be taken into account. During the oxidation of the glucose, we have two main metabolic pathways involved and both have a net effect over the proton flux:
-
- As we can observe, the production of protons by oxidative phosphorylation is three times higher than the one produced by the glycolysis per molecule of glucose. However, the chemical rate of production could be used to determine which is the main source of protons in our sample under specific conditions.
- We have also to take into account the pka for the point of equilibrium of the most common weak acids that will be formed during both processes:
Additional resources
» O2k-Manual: MiPNet23.15 O2k-pH ISE-Module
» O2k-SOP: MiPNet08.16 pH calibration
» MitoPedia O2k and high-resolution respirometry: O2k hardware
MitoPedia O2k and high-resolution respirometry:
O2k-Open Support
O2k signal and output
- O2k signal: The O2k-pH ISE-Module is operated through the pX channel of the O2k, with electric potential (volt [V]) as the primary and raw signal
- O2k output: type I and II
pH changes versus glycolytic flux
- Measurement of extracellular proton flux and glycolytic flux are related under specifically conrolled conditions. Such conditions must be carefully evaluated, may require modifications of protocols, and need data analysis beyond reporting changes of pH.
- The extracellular acidification rate (ECAR) is the change of pH over time, which may be of interest in relation to acidification problems in a culture medium or incubation buffer. pH is the negative logarithm of proton activity. Comparable to volume-specific [[oxygen flux] [pmol·s-1·mL-1]], which is the (negative) time derivative of oxygen concentration measured in a closed system, volume-specific proton flux is the time derivative of proton concentration, expressed in units [pmol·s-1·mL-1]]. The physiologically relevant metabolic proton flux, therefore, must not be confused with ECAR.
- » Proton flux
- To accurately measure biologically induced changes in pH, the buffering capacity of the medium has to be small. This may be addressed either by using or preparing media with a buffering capacity that is low but still sufficient to keep the pH in the desired range for a limited period of time. An alternative approach is to use buffers with very low buffering capacity and keep the pH value inside the desired limits by a pH-Stat.
Compare measurement of pH with the pH electrode and ratiometric fluorometric methods (NextGen-O2k)
- » Carboxy SNARF 1
- » HPTS
O2k-Manual
Introduction and scope
- The Oroboros O2k supports the modular O2k-MultiSensor extension for recording potentiometric (voltage) signals simultaneously with the oxygen signals in both O2k-chambers. Potentiometric measurements result in a voltage signal (pX) which is typically a linear function of the logarithm of the activity (concentration) of the substance of interest (the analyte). A calibrated pH electrode displays the negative decadic logarithm of the H+ ion activity (potentia hydrogenii) and thus got its name “pH electrode”. Using a pH / reference electrode module,the extracellular proton flux can either be calculated by changes in pH over time (previous calculation of buffering capacity of the medium required) or by the amount of injected base via pH Stat. MiPNet 16.0x
DatLab analysis
pX reference layouts
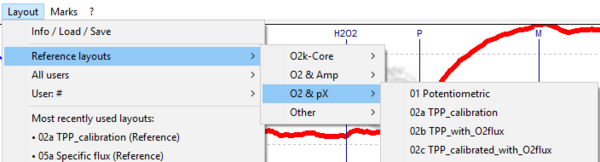
- Graph layout: Four reference layouts are available in DatLab 7 based on the recorded pX signal:
- 01 Potentiometric
- 02a TPP_calibration
- 02b TPP_with_O2flux
- 02c TPP_calibrated_with_O2flux
- These layouts can be selected in [Layout / Reference layouts / O2 & pX].
- Graph layout: Four reference layouts are available in DatLab 7 based on the recorded pX signal:
- Reference layouts can be modified and saved as user-defined layouts, see MitoPedia: DatLab.
pX settings
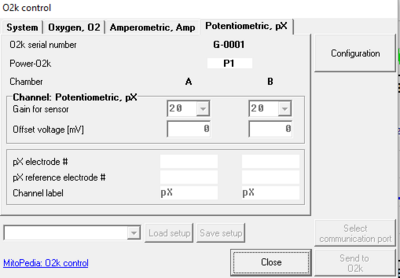
- In the O2k configuration window the pX channel can be activated and a label for the inserted pX electrode can be entered for documentation purposes.
- Gain and Offset voltage [mV] for the pX channel can be set in the O2k control window [F7], tab: Potentiometric, pX. The gain influences the "pX Raw Signal” recorded in DatLab. Therefore, a gain of 1 will give the same voltage [V] as would be measured with any multimeter between reference and measuring electrode.
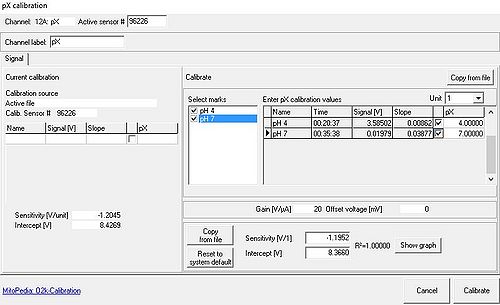
pX calibration
- From the Calibration menu the pX Calibration window window [Potentiometric, pX] is opened. This feature allows for a simple two-point linear calibration of pX (or –pX) as a function of recorded voltage, using data ranges marked for calibration [Select marks] or known pX values, see O2k-calibration and pH calibration.
- Calibrations for different signal types: There is only one set of calibration values for each pX channel, irrespective of the connected electrode. If a pX channel was calibrated for a pH electrode, these values will initially also be used to calculate the calibrated signal when the pH electrode is exchanged for a TPP+ electrode. Even when observing only the raw (not the calibrated) signal, the time derivative (Slope pX) will be calculated from the calibrated signal, which might lead to confusion when the time derivative is used to access stability or signal drift. It is, therefore, suggested to set the calibrated signal to the raw signal whenever the raw signal is to be used as the primary data source.
- Calibration values from other files can be imported with Copy from file in the pX calibration window.
Calculate proton production in DatLab
- Note: Requires DatLab 5 or DatLab 6
- Proton production rates can be calculated in real-time during data acquisition. Select the menu [Flux/Slope]/[Proton Flux].
- Determining the buffering capacity of the medium:
- Calibrate the pH electrode, observe the calibrated pH signal.
- Fill TIP syringes with diluted acid or base.
- Start a slow injection of acid or base into the media (no sample present).
- Place a mark on a stable region of the slope plot of the calibrated pH signal 'pX slope".
- Go to [Plots]/[Proton Flux].
- The buffering capacity is calculated by DatLab and can be used for calculations of biological proton flow in the same file or noted down and used in subsequent experiments.
- Biological proton flux:
- Calibrate the pH electrodes.
- Observe the pH calibrated signal.
- Place marks on regions of interest on the pX slope plot. If you use the "pH" stat" se above to keep the pH value in desired limit make sure that you exclude times during which base was injected.
- Select [Flux/Slope] / [Proton Flux].
- Enter the buffering capacity in the appropriate field or use the feature in the upper part of the window to calculate the buffering capacity from a calibration experiment in the same file.
- Press [OK].
- A new plot "Proton Flux" is now available in [Graph]/[Select Plots] (right at the end of the list. You can now chose to display this plot e.g. instead of the pX slope plot by selecting its check box and de-selecting the check box of the "pX slope" plot.
- Determining the buffering capacity of the medium:
- Proton production rates can be calculated in real-time during data acquisition. Select the menu [Flux/Slope]/[Proton Flux].
- Known issues: DatLab always calculates a new buffering capacity from the input in the upper part of the window and does not remember the value from previous files. Therefore, if the determination of buffering capacity was done in a different Datlab file the value has to manually entered.
Specifications
- Specifications provided by OROBOROS INSTRUMENTS for quality control of pH electrodes:
- Drift (after 45 min stabilization, integrated over 5 minutes, 37 °C, 2 mM buffering capacity): <= 20 µpH/s.
- Specifications provided by OROBOROS INSTRUMENTS for quality control of pH electrodes:
pH-Stat
- TIP2k setups in the DatLab template file DLTemplates_pH.dlt and Spreadsheet (e.g. Excel) templates for determining proton form base injections are available for download: File:DLTemplates.dlt. Please note that of course the more straightforward calculation of proton flows from the measured pH slope is also possible while operating in pH-Stat mode!
Applications
- For simultaneous measurement of O2 and pH, we refer to the classical literature on bioenergetics and the discovery of the chemiosmotic coupling mechanism, the quantification of H+/O2 stoichiometric ratios for proton pumping (Peter Mitchell). Other groups (e.g. SE_Lund_Elmer E) have used the pH electrode in the O2k in conjunction with a study of mitochondrial permeability transition.
- The majority of novel applications will address the problem of aerobic glycolysis in intact cells, using the measurement of proton production as an indirect but continuous record of lactate production and corresponding acidification of the medium, while simultaneously monitoring oxygen concentration and oxygen consumption. In a well buffered culture medium, the pH change is extremely small relative to the amount of protons (lactic acid) produced, hence a low-buffering capacity medium needs to be applied. A titration of acid (lactic acid or HCl) into the low-buffering capacity medium yields the pH-dependent buffering capacity (Delta H+ added/Delta H+ measured by the pH electrode). Under various metabolic conditions, lactic acid production is the dominant mechanism causing acidification, hence the pH measurement is a good indirect indicator of aerobic glycolysis.
Protocols
The SUIT-003 O2 ce D067 is designed to study the effect of glucose (or fructose; Crabtree effect) on ROUTINE respiration and glycolysis inhibition on LEAK-respiration simultaneously measured with proton flux in living cells. If the aim is to analyze only the Crabtree effect and the effect of 2-Deoxyglucose on proton flux and respiration via glycolysis inhibition, Oligomycin can be omitted. If there is an interest in studying coupling control, oligomycin should be added to promote LEAK-respiration. To obtain more information about the Crabtree effect, it is ideal to use in parallel the protocol SUIT-003 O2 ce D038 (glucose addition in ET state) and SUIT-003 O2 ce D037 (glucose addition in ROUTINE). The latter can be extended by the cell viability test and the Complex IV module.
Demo Experiment with simulated proton flow
- Medium: imidazole buffered medium, see above
- Calibration steps (for calculating buffering capacity): 30, 90, 150 HCl pmol/(s mL): TIP 1 mmol/L HCl, pump speed 0.06, 0.18, 0.3 µL/s
- Simulated proton flow 30,90,150 pmol HCl/(s mL) in pH stat mode: The pH value was held inside narrow limits by using the TIP in pH stat mode (100 mM KOH, Tip set up similar to the one included in DLTemplates_pH.dlt available from http://www.oroboros.at/index.php?id=ph-oxygen. The proton flow was simulated using a second TIP
- Bioblast links: pH and protons - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- pH and protons
- » pH
- » hydrogen ion H+
- » hydron H+
- » hydronium ion H3O+
- » hydride H-
- » proton p+
- » pH buffering capacity
- » proton flux
- » proton pump versus hydrogen ion pump
- » proton leak
- » proton slip
- » protonmotive force
- pH and protons
- O2k-pH
- » O2k-Catalogue: O2k-pH ISE-Module
- » O2k-Manual pH electrode: MiPNet23.15 O2k-pH ISE-Module
- » O2k-SOP: MiPNet08.16 pH calibration
- » File:PH-Calibration-List.xls
- » NextGen-O2k, ratiometric: Carboxy SNARF 1
- » NextGen-O2k, ratiometric: HPTS
- » pH calibration buffers
- O2k-pH
- O2k-Publications
- HRFR - general
- » O2k-Manual: MiPNet22.11 O2k-FluoRespirometer manual
- » O2k signals and output
- » O2k-SOP: MiPNet14.06 Instrumental O2 background
- » MiPNet19.18A O2k-Series G: Start
- » ESD
- » O2k configuration
- » O2k control
- » O2k-FluoRespirometer
- » O2k-Main Unit#O2k-Series
- » Titration-Injection microPump
- » Compare: O2k-TPP+_ISE-Module
- HRFR - general
- DatLab
References
- O2k-Manual: Contents: O2k-Core Manual.pdf
| Chapter | Section | Last update |
|---|---|---|
| MiPNet07.08 User information | O2k-Manual: user information. PLEASE STUDY THIS MANUAL. | 2017-11-03 |
| MiPNet19.18 O2k-Series G Core manual | O2k-Core manual contents. | 2016-08-08 |
| MiPNet19.18B POS-service | Service of the polarographic oxygen sensor OroboPOS. | 2021-06-23 |
| MiPNet19.18C DatLab 6: Guide | DatLab-guide through the menus. | 2016-08-24 |
| MiPNet19.18E O2 flux analysis - DatLab 6 | Oxygen flux analysis: DatLab real-time. | 2016-08-08 |
| MiPNet26.06 DatLab 7: Guide | DatLab-guide through the menus. | 2021-06-02 |
| MiPNet28.10 SmartPOS-service | SmartPOS: Service | 2023-09-29 |
- TIP2k-Manual***
- » O2k-Catalogue: TIP2k
- » O2k-Publications: TIP2k
| Chapter | Section | Last update |
|---|---|---|
| MiPNet12.10 TIP2k-manual | Titration-Injection microPump TIP2k manual. | 2021-07-08 |
- Bioblast links: pH and protons - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- pH and protons
- » pH
- » hydrogen ion H+
- » hydron H+
- » hydronium ion H3O+
- » hydride H-
- » proton p+
- » pH buffering capacity
- » proton flux
- » proton pump versus hydrogen ion pump
- » proton leak
- » proton slip
- » protonmotive force
- pH and protons
- O2k-pH
- » O2k-Catalogue: O2k-pH ISE-Module
- » O2k-Manual pH electrode: MiPNet23.15 O2k-pH ISE-Module
- » O2k-SOP: MiPNet08.16 pH calibration
- » File:PH-Calibration-List.xls
- » NextGen-O2k, ratiometric: Carboxy SNARF 1
- » NextGen-O2k, ratiometric: HPTS
- » pH calibration buffers
- O2k-pH
- O2k-Publications
- HRFR - general
- » O2k-Manual: MiPNet22.11 O2k-FluoRespirometer manual
- » O2k signals and output
- » O2k-SOP: MiPNet14.06 Instrumental O2 background
- » MiPNet19.18A O2k-Series G: Start
- » ESD
- » O2k configuration
- » O2k control
- » O2k-FluoRespirometer
- » O2k-Main Unit#O2k-Series
- » Titration-Injection microPump
- » Compare: O2k-TPP+_ISE-Module
- HRFR - general
- DatLab
MitoPedia concepts:
MiP concept
MitoPedia methods:
Respirometry,
Fluorimetry