Difference between revisions of "Chinopoulos 2012 Abstract Bioblast"
| Line 5: | Line 5: | ||
|year=2012 | |year=2012 | ||
|event=[[Bioblast 2012]] | |event=[[Bioblast 2012]] | ||
|abstract=SUCLA2 encodes for the ADP-forming β-subunit (A-SUCL-β) of succinyl | |abstract=[[File:Christos Chinopoulos.jpg|right|150px|Christos Chinopoulos]] | ||
SUCLA2 encodes for the ADP-forming β-subunit (A-SUCL-β) of succinyl | |||
CoA ligase, an enzyme of the citric acid cycle. Mutations in SUCLA2 lead | CoA ligase, an enzyme of the citric acid cycle. Mutations in SUCLA2 lead | ||
to a mitochondrial disorder associated with mitochondrial DNA depletion. | to a mitochondrial disorder associated with mitochondrial DNA depletion. | ||
| Line 25: | Line 26: | ||
by mutations in this gene originate solely from the neuronal cell | by mutations in this gene originate solely from the neuronal cell | ||
population. | population. | ||
|keywords=SUCLA2, Mitochondrial disorder, Cortex, | |keywords=SUCLA2, Mitochondrial disorder, Cortex, | ||
|mipnetlab=HU Budapest Chinopoulos C, | |mipnetlab=HU Budapest Chinopoulos C, | ||
|journal=Mitochondr Physiol Network | |journal=Mitochondr Physiol Network | ||
|articletype=Abstract | |articletype=Abstract | ||
| Line 38: | Line 39: | ||
}} | }} | ||
__NOTOC__ | __NOTOC__ | ||
== Affiliations and author contributions == | == Affiliations and author contributions == | ||
| Line 53: | Line 53: | ||
(5) Department of Medical Biochemistry, Semmelweis University, Hungary | (5) Department of Medical Biochemistry, Semmelweis University, Hungary | ||
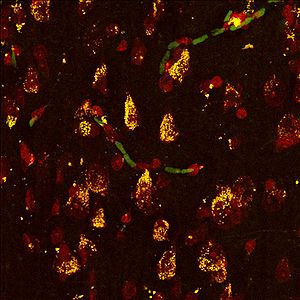
== Figure 1 == | |||
[[File:Christos Nissl Sucla2.jpg|300px|Nissl Sucla2]] | |||
Co-localization of fluorescent Nissl staining the cytosol of neurons (red) | |||
and the ADP-forming β-subunit (A-SUCL-beta) | |||
of succinyl CoA ligase (green-->yellow) in human temporal cortex. | |||
== Help == | == Help == | ||
* [[Abstracts help]] | * [[Abstracts help]] | ||
* [[MitoPedia Glossary: Terms and abbreviations]] | * [[MitoPedia Glossary: Terms and abbreviations]] | ||
Revision as of 09:17, 15 November 2012
| Dobolyi A, Ostergaard E, Bagó AG, Palkovits M, Adam-Vizi V, Chinopoulos C (2012) Exclusive neuronal expression of SUCLA2 in the human brain. Mitochondr Physiol Network 17.12. |
Link: MiPNet17.12 Bioblast 2012 - Open Access
Chinopoulos C (2012)
Event: Bioblast 2012
SUCLA2 encodes for the ADP-forming β-subunit (A-SUCL-β) of succinyl CoA ligase, an enzyme of the citric acid cycle. Mutations in SUCLA2 lead to a mitochondrial disorder associated with mitochondrial DNA depletion. This mitochondrial disorder manifests as neonatal encephalomyopathy exhibiting dystonia, deafness and pronounced lesions in the basal ganglia. Despite that a SUCLA2 gene defect results in distinct brain pathology, precise localization of the encoded protein has never been investigated. Here we show that the immunoreactivity of A-SUCL-β in the human cerebral cortex was present exclusively in neurons, identified by their morphology and visualized by double labelling with a fluorescent Nissl dye. The A-SUCL-β immunoreactivity co-localized >99% with that of the d-subunit of the mitochondrial F0-F1 ATP synthase. Specificity of the anti-A-SUCL-β antiserum was verified by the absence of labelling in fibroblasts from a patient with a complete deletion of SUCLA2. A-SUCL-β immunoreactivity was absent in glial cells, identified by antibodies directed against the glial markers GFAP and S100. Our work establishes that SUCLA2 is expressed exclusively in neurons in the human cerebral cortex. Therefore, all encephalopathic features of the disease emerging by mutations in this gene originate solely from the neuronal cell population.
• Keywords: SUCLA2, Mitochondrial disorder, Cortex
• O2k-Network Lab: HU Budapest Chinopoulos C
Labels:
Stress:Mitochondrial Disease; Degenerative Disease and Defect"Mitochondrial Disease; Degenerative Disease and Defect" is not in the list (Cell death, Cryopreservation, Ischemia-reperfusion, Permeability transition, Oxidative stress;RONS, Temperature, Hypoxia, Mitochondrial disease) of allowed values for the "Stress" property. Organism: Human Tissue;cell: Neurons; Brain"Neurons; Brain" is not in the list (Heart, Skeletal muscle, Nervous system, Liver, Kidney, Lung;gill, Islet cell;pancreas;thymus, Endothelial;epithelial;mesothelial cell, Blood cells, Fat, ...) of allowed values for the "Tissue and cell" property.
Affiliations and author contributions
Arpád Dobolyi (1), Elsebet Ostergaard (2), Attila G. Bagó (1,3), Miklós Palkovits (1,4), Vera Adam-Vizi (5) and Christos Chinopoulos (5)
(1) Department of Anatomy, Histology and Embryology, Semmelweis University, Hungary; Email: [email protected]
(2) Department of Clinical Genetics, Copenhagen University Hospital Rigshospitalet, Denmark
(3) National Institute of Neurosurgery, Hungary
(4) Human Brain Tissue Bank, Semmelweis University, Hungary
(5) Department of Medical Biochemistry, Semmelweis University, Hungary
Figure 1
Co-localization of fluorescent Nissl staining the cytosol of neurons (red)
and the ADP-forming β-subunit (A-SUCL-beta)
of succinyl CoA ligase (green-->yellow) in human temporal cortex.