Bouillaud 2012 Abstract Bioblast
| Bouillaud F (2012) Sulfide - a remarkable mitochondrial substrate. Mitochondr Physiol Network 17.12. |
Link: MiPNet17.12 Bioblast 2012 - Open Access
Bouillaud F (2012)
Event: Bioblast 2012
Sulfide (H2S gas, HS- anion) shows the same toxicity as cyanide, NO or CO for Complex IV (cytochrome oxidase; CIV). NO, CO and H2S are considered as gasotransmitters of physiological relevance. While signaling is expected to occur by different pathways, the question of involvement of mitochondria remains sometimes unresolved and particularly because some experiments used concentrations relevant to bioenergetics. Cellular metabolism generates low amount of sulfide and the activity of bacteria in the colonic lumen exposes the colonic wall to extracellular concentrations of sulfide [#60 µM], large enough to inhibit cellular respiration. Therefore, the question of sulfide disposal needs to be adressed. Mitochondria themselves appear as best candidate to explain sulfide disposal: a Sulfide Oxidation Unit (SOU) oxidizes sulfide into thiosulfate in many cell types in culture and in mitochondria from liver, heart or kidney. When sulfide is infused to mitochondria or cells at rates that stay within their sulfide oxidation capacities they oxidize it and maintain a low (<500 nM with cells) external concentration of sulfide well behind the toxic level (IC50 10-20 µM with cells). Notably, SOU activity could not be detected in brain mitochondria or neuroblastoma cells making them intolerant to sulfide. SOU is constituted by a sulfide quinone reductase (SQR) associated with a sulfur transferase and a dioxygenase. Present knowledge indicates that two molecules of H2S and one of oxygen (O2) are consumed by SOU to deliver two electrons to quinone. Then the stoichiometry for mitochondrial respiration based on sulfide oxidation is (1+0.5) O2 / 2 H2S = 0.75 and for the same rate of electron transfer three times more oxygen are needed than with NADH/FADH2 coenzymes. In consequence, infusing sulfide immediately and significantly increases oxygen consumption of respiring cells/mitochondria.
Mitochondria show a high affinity for sulfide and its oxidation usually takes priority over ongoing mitochondrial oxidation processes. Something that is mandatory to ensure efficient sulfide disposal in presence of largely greater intracellular concentrations of other substrates. Extracellular sulfide is therefore a remarkable substrate: it could be used at nanomolar concentrations, its gaseous nature allows fast transfer to mitochondria without transporters, it is directly usable to reduce quinone without metabolic processing (including for example initial ATP consuming steps). This metabolic role of sulfide and its reductant properties contrast sharply with the other two gazotransmitters and particularly with NO which is pro-oxidant. There are still uncertainties about the lowest concentration of sulfide that SQR would detect but, in physiological conditions, the overlap between bioenergetically relevant and signaling concentrations appears even more likely with sulfide than with NO or CO.
Colonocytes are adapted to high sulfide exposure. In these cells reverse bioenergetic reactions including reverse electron flux in Complex I are taking place to ensure continuation of a fast sulfide oxidation even if CIV is inhibited.
• Keywords: Sulfide, Gasotransmitters, CHO cells, Colonocytes
• O2k-Network Lab: FR Paris Bouillaud F
Labels: MiParea: Respiration
Organism: Human
Tissue;cell: CHO
Enzyme: Complex I, Complex IV;cytochrome c oxidase Regulation: Coupling efficiency;uncoupling, Inhibitor
Pathway: N, CIV, Other combinations HRR: Oxygraph-2k, TIP2k
Affiliations and author contributions
Mitochondries, bioenergetique, metabolisme et signalisation - Institut Cochin, INSERM U567, CNRS UMR 8104 - Universite Paris Descartes, Departement d'Endocrinologie, Métabolisme et Cancer 24, Rue du Faubourg Saint-Jacques 75 014 Paris, FRANCE; E-mail: [email protected]
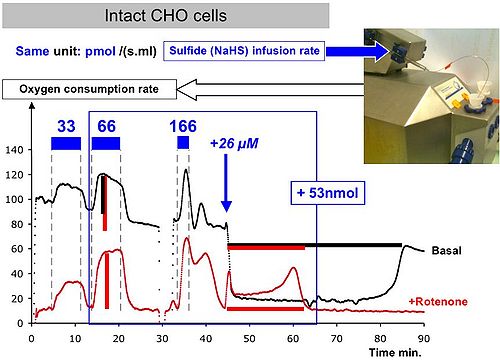
Figure 1
The oxygen consumption (JO2) of CHO cells in suspension is recorded, the background rate (black) and in presence of rotenone inhibition of mitochondrial Complex I (red) are shown. Sulfide additions are indicated in blue. Addition of the same amount of sulfide (+53 nmol in 2 ml) has different consequences when it is infused (blue bars) at a rate that matches with the sulfide oxidation capacities of cells (66), exceeds it (166) or added as a single injection (+26 µM). Note that the maximal sulfide flux (66) compares well with the initial background JO2 (100). The increase in JO2 is figured with vertical bars (red/black) with the red repeated side to the black for comparison. The ratio (increase in JO2) / (sulfide injection rate) provides an indication of the stoichiometry oxygen / sulfide. This value is close to the theorical value of 0.75 in presence of rotenone but close to 0.5 for background. This suggests that electrons from sulfide replaced electrons previously provided to quinone by Complex I (priority of sulfide oxidation over other substrates). In contrast, when inhibition by sulfide takes place (+26 µM) the length of the horizontal red/black bars (= time to oxidize sulfide) indicates that under these conditions Complex I becomes an opponent to sulfide oxidation probably because of the change in its reduction state.