Shadel 2005 Trends Biochem Sci: Difference between revisions
From Bioblast
(Created page with "{{Publication |title=Shadel GS (2005) Mitochondrial DNA, aconitase 'wraps' it up. Trends Biochem Sci 30:294-6. https://doi.org/10.1016/j.tibs.2005.04.007 |info=[https://pubmed...") |
No edit summary |
||
| Line 8: | Line 8: | ||
|editor=Gnaiger E | |editor=Gnaiger E | ||
}} | }} | ||
[[File:Shadel 2005 Trends Biochem Sci CORRECTION.png|right|300px]] | [[File:Shadel 2005 Trends Biochem Sci CORRECTION.png|right|300px]] | ||
{{Template:Correction FADH2 and S-pathway}} | {{Template:Correction FADH2 and S-pathway}} | ||
Latest revision as of 02:54, 5 November 2023
| Shadel GS (2005) Mitochondrial DNA, aconitase 'wraps' it up. Trends Biochem Sci 30:294-6. https://doi.org/10.1016/j.tibs.2005.04.007 |
Shadel GS (2005) Trends Biochem Sci
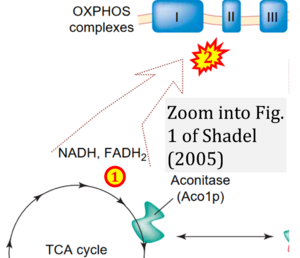
Abstract: Mitochondria are the sites of many essential biochemical reactions, an important subset of which require proteins encoded in the mitochondrial DNA (mtDNA). How mtDNA is regulated in response to changing cellular demands is largely unknown. A recent study documents that the mitochondrial TCA-cycle enzyme aconitase is associated with protein-mtDNA complexes called nucleoids. In this novel context, aconitase functions to stabilize mtDNA, perhaps by reversibly remodeling nucleoids to directly influence mitochondrial gene expression in response to changing cellular metabolism.
• Bioblast editor: Gnaiger E
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«