Difference between revisions of "Posch 2022 MitoFit"
Tindle Lisa (talk | contribs) (Created page with "{{MitoFit page name}} {{Publication |title=Posch P, Dichtl S, Zaderer V, Lass-Flörl C, Wilfingseder D (2022) How to optimize respiratory models for SARS-CoV-2 research. Mito...") |
|||
| Line 1: | Line 1: | ||
{{Publication | {{Publication | ||
|title=Posch P, Dichtl S, Zaderer V, Lass-Flörl C, Wilfingseder D (2022) How to optimize respiratory models for SARS-CoV-2 research. MitoFit Preprints 2022.4. https://doi.org/10.26124/mitofit:2022-0004 | |title=Posch P, Dichtl S, Zaderer V, Lass-Flörl C, Wilfingseder D (2022) How to optimize respiratory models for SARS-CoV-2 research. MitoFit Preprints 2022.4. https://doi.org/10.26124/mitofit:2022-0004 | ||
|info=[[File:MitoFit Preprints pdf.png|left|160px|link=https://wiki.oroboros.at/images/e/e5/Posch_2022_MitoFit.pdf|MitoFit pdf]] [https://wiki.oroboros.at/images/e/e5/Posch_2022_MitoFit.pdf How to optimize respiratory models for SARS-CoV-2 research]<br/> | |info=[[File:MitoFit Preprints pdf.png|left|160px|link=https://wiki.oroboros.at/images/e/e5/Posch_2022_MitoFit.pdf|MitoFit pdf]] [https://wiki.oroboros.at/images/e/e5/Posch_2022_MitoFit.pdf How to optimize respiratory models for SARS-CoV-2 research]<br/> | ||
|authors=Posch Wilfried, Dichtl Stefanie, Zaderer Viktoria, Lass-Floerl Cornelia, Wilflingseder Doris | |authors=Posch Wilfried, Dichtl Stefanie, Zaderer Viktoria, Lass-Floerl Cornelia, Wilflingseder Doris | ||
|year=2022-03-24 | |year=2022-03-24 | ||
|journal=MitoFit Prep | |journal=MitoFit Prep | ||
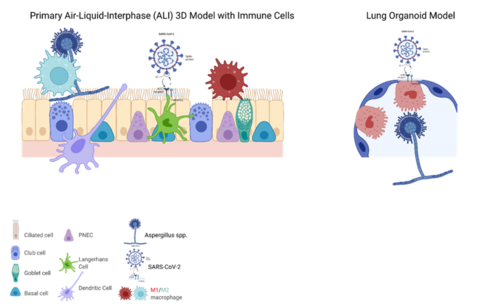
|abstract= | |abstract=[[File:Posch 2022 MitoFit Graphical abstract.png|right|500px|Graphical abstract]] Sophisticated 3D cell culture tissue models experienced a boom in the last years and in particular human cell culture and 3D respiratory systems greatly supported the development of novel drugs and vaccines during the SARS-CoV-2 pandemic lately. These models provide multiple benefits in terms of similarities in differentiation, metabolism, receptor expression, polarity, infectivity compared to human tissues and thus provide excellent models to study very first interactions with the host during pathogen entry. Dependent on the experimental approach, the use of different 3D models is more beneficial – apical out lung organoids for e.g., high content screening (HCS) of treatment options, air-liquid interphase (ALI) models for e.g., easy incorporation of immune cells, screening of epithelial integrity or mucociliary clearance. This review will give an overview on the models established in our laboratory and on their applications. <br><br> | ||
[[File:Posch 2022 MitoFit Graphical abstract.png|right|500px|Graphical abstract]] Sophisticated 3D cell culture tissue models experienced a boom in the last years and in particular human cell culture and 3D respiratory systems greatly supported the development of novel drugs and vaccines during the SARS-CoV-2 pandemic lately. These models provide multiple benefits in terms of similarities in differentiation, metabolism, receptor expression, polarity, infectivity compared to human tissues and thus provide excellent models to study very first interactions with the host during pathogen entry. Dependent on the experimental approach, the use of different 3D models is more beneficial – apical out lung organoids for e.g., high content screening (HCS) of treatment options, air-liquid interphase (ALI) models for e.g., easy incorporation of immune cells, screening of epithelial integrity or mucociliary clearance. This review will give an overview on the models established in our laboratory and on their applications. <br><br> | |keywords=respiratory models, air-liquid interphase,SARS-CoV-2 | ||
|keywords= | |||
respiratory models, air-liquid interphase,SARS-CoV-2 | |||
|editor=Tindle-Solomon L | |editor=Tindle-Solomon L | ||
}} | }} | ||
{{Labeling | {{Labeling | ||
|diseases=Infectious | |||
|diseases= | |||
|organism=Human | |organism=Human | ||
|tissues=Lung;gill, Endothelial;epithelial;mesothelial cell, Lymphocyte | |||
|additional=organoid, 3D model | |||
}} | }} | ||
{{MitoFit page name}} | |||
ORC'''ID''': [[File:ORCID.png|20px|link=https://orcid.org/0000-0001-8955-7654]] Posch Wilfried, [[File:ORCID.png|20px|link=https://orcid.org/0000-0002-7657-329X]] Dichtl Stefanie, [[File:ORCID.png|20px|link=https://orcid.org/0000-0002-0507-0513]] Zaderer Viktoria, [[File:ORCID.png|20px|link=https://orcid.org/0000-0002-2946-7785]] Lass-Flörl Cornelia, [[File:ORCID.png|20px|link=https://orcid.org/0000-0002-5888-5118]] Wilflingseder Doris | |||
Revision as of 15:18, 24 March 2022
| Posch P, Dichtl S, Zaderer V, Lass-Flörl C, Wilfingseder D (2022) How to optimize respiratory models for SARS-CoV-2 research. MitoFit Preprints 2022.4. https://doi.org/10.26124/mitofit:2022-0004 |
»
How to optimize respiratory models for SARS-CoV-2 research
Posch Wilfried, Dichtl Stefanie, Zaderer Viktoria, Lass-Floerl Cornelia, Wilflingseder Doris (2022-03-24) MitoFit Prep
Abstract:
Sophisticated 3D cell culture tissue models experienced a boom in the last years and in particular human cell culture and 3D respiratory systems greatly supported the development of novel drugs and vaccines during the SARS-CoV-2 pandemic lately. These models provide multiple benefits in terms of similarities in differentiation, metabolism, receptor expression, polarity, infectivity compared to human tissues and thus provide excellent models to study very first interactions with the host during pathogen entry. Dependent on the experimental approach, the use of different 3D models is more beneficial – apical out lung organoids for e.g., high content screening (HCS) of treatment options, air-liquid interphase (ALI) models for e.g., easy incorporation of immune cells, screening of epithelial integrity or mucociliary clearance. This review will give an overview on the models established in our laboratory and on their applications.
• Keywords: respiratory models, air-liquid interphase, SARS-CoV-2 • Bioblast editor: Tindle-Solomon L
Labels:
Pathology: Infectious
Organism: Human Tissue;cell: Lung;gill, Endothelial;epithelial;mesothelial cell, Lymphocyte
organoid, 3D model
Posch 2022 MitoFit
ORCID: ![]() Posch Wilfried,
Posch Wilfried, ![]() Dichtl Stefanie,
Dichtl Stefanie, ![]() Zaderer Viktoria,
Zaderer Viktoria, ![]() Lass-Flörl Cornelia,
Lass-Flörl Cornelia, ![]() Wilflingseder Doris
Wilflingseder Doris