Pelletier-Galarneau 2021 Curr Cardiol Rep: Difference between revisions
No edit summary |
No edit summary |
||
| Line 10: | Line 10: | ||
|editor=Gnaiger E | |editor=Gnaiger E | ||
}} | }} | ||
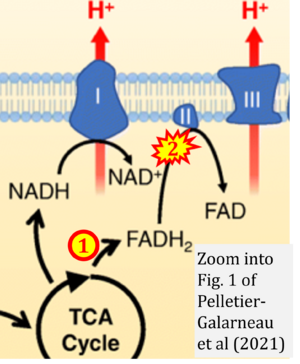
[[File:Pelletier-Galarneau 2021 Curr Cardiol Rep CORRECTION.png|right| | [[File:Pelletier-Galarneau 2021 Curr Cardiol Rep CORRECTION.png|right|300px]] | ||
{{Template:Correction FADH2 and S-pathway}} | {{Template:Correction FADH2 and S-pathway}} | ||
{{Labeling | {{Labeling | ||
|enzymes=Complex II;succinate dehydrogenase | |enzymes=Complex II;succinate dehydrogenase | ||
}} | }} | ||
Latest revision as of 12:15, 15 October 2023
| Pelletier-Galarneau M, Detmer FJ, Petibon Y, Normandin M, Ma C, Alpert NM, El Fakhri G (2021) Quantification of myocardial mitochondrial membrane potential using PET. Curr Cardiol Rep 23:70. https://doi.org/10.1007/s11886-021-01500-8 |
Pelletier-Galarneau M, Detmer FJ, Petibon Y, Normandin M, Ma C, Alpert NM, El Fakhri G (2021) Curr Cardiol Rep
Abstract: Purpose of review: To present a method enabling in vivo quantification of tissue membrane potential (ΔΨT), a proxy of mitochondrial membrane potential (ΔΨm), to review the origin and role of ΔΨm, and to highlight potential applications of myocardial ΔΨT imaging.
Recent findings: Radiolabelled lipophilic cations have been used for decades to measure ΔΨm in vitro. Using similar compounds labeled with positron emitters and appropriate compartment modeling, this technique now allows in vivo quantification of ΔΨT with positron emission tomography. Studies have confirmed the feasibility of measuring myocardial ΔΨT in both animals and humans. In addition, ΔΨT showed very low variability among healthy subjects, suggesting that this method could allow detection of relatively small pathological changes. In vivo assessment of myocardial ΔΨT provides a new tool to study the pathophysiology of cardiovascular diseases and has the potential to serve as a new biomarker to assess disease stage, prognosis, and response to therapy.
• Bioblast editor: Gnaiger E
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Labels:
Enzyme: Complex II;succinate dehydrogenase