Lewis 2019 Int J Mol Sci
| Lewis MT, Kasper JD, Bazil JN, Frisbee JC, Wiseman RW (2019) Quantification of mitochondrial oxidative phosphorylation in metabolic disease: application to Type 2 diabetes. Int J Mol Sci 20:5271. |
Lewis MT, Kasper JD, Bazil JN, Frisbee JC, Wiseman RW (2019) Int J Mol Sci
Abstract: Type 2 diabetes (T2D) is a growing health concern with nearly 400 million affected worldwide as of 2014. T2D presents with hyperglycemia and insulin resistance resulting in increased risk for blindness, renal failure, nerve damage, and premature death. Skeletal muscle is a major site for insulin resistance and is responsible for up to 80% of glucose uptake during euglycemic hyperglycemic clamps. Glucose uptake in skeletal muscle is driven by mitochondrial oxidative phosphorylation and for this reason mitochondrial dysfunction has been implicated in T2D. In this review we integrate mitochondrial function with physiologic function to present a broader understanding of mitochondrial functional status in T2D utilizing studies from both human and rodent models. Quantification of mitochondrial function is explained both in vitro and in vivo highlighting the use of proper controls and the complications imposed by obesity and sedentary lifestyle. This review suggests that skeletal muscle mitochondria are not necessarily dysfunctional but limited oxygen supply to working muscle creates this misperception. Finally, we propose changes in experimental design to address this question unequivocally. If mitochondrial function is not impaired it suggests that therapeutic interventions and drug development must move away from the organelle and toward the cardiovascular system.
• Bioblast editor: Gnaiger E • O2k-Network Lab: US MI East Lansing Bazil JN
High-resolution respirometry
- "Instruments used to measure respiration in isolated mitochondria primarily include Oroboros O2k (Oroboros Instruments) and Seahorse XF Analyzers (Agilent). In the present review, the Oroboros O2k is primarily focused on because of its advantage in accuracy relative to Seahorse XF Analyzers which are designed for high-throughput qualitative measurements [51]" (page 6).
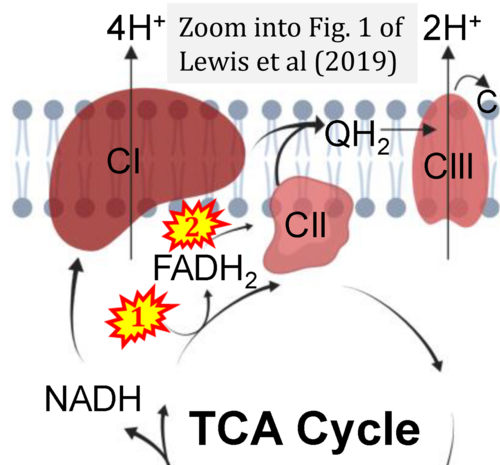
Correction: FADH2 and S-pathway
- A commonly found error on FADH2 in the S-pathway requires correction. For clarification, see page 48 in Gnaiger (2020)
- Quote (p 48): "The substrate of CII is succinate, which is oxidized forming fumarate while reducing flavin adenine dinucleotide FAD to FADH2, with further electron transfer to the quinone pool. Whereas reduced NADH is a substrate of Complex I linked to dehydrogenases of the TCA cycle and mt-matrix upstream of CI, reduced FADH2 is a product of Complex II with downstream electron flow from CII to Q."
- A commonly found error on FADH2 in the S-pathway requires correction. For clarification, see page 48 in Gnaiger (2020)
- Gnaiger E (2020) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 5th ed. Bioenerg Commun 2020.2. https://doi.org/10.26124/bec:2020-0002
Labels: MiParea: Respiration, Exercise physiology;nutrition;life style, mt-Medicine
Pathology: Diabetes
Organism: Human, Rat Tissue;cell: Skeletal muscle
Regulation: Cyt c, Oxygen kinetics, PCr;Cr
Coupling state: OXPHOS
HRR: Oxygraph-2k
BMI