Ingram 2020 MitoFit Preprint Arch: Difference between revisions
No edit summary |
No edit summary |
||
| Line 16: | Line 16: | ||
== Figure == | == Figure == | ||
[[File:Chakrabarti Figure 1.png|1250px]] | [[File:Chakrabarti Figure 1.png|1250px]] | ||
[[File:Chakrabarti Figure 2.png|1250px]] | |||
== References == | == References == | ||
:::# Zhang J, Wei L, Hu X, Xie B, Zhang Y, Wu G-R, et al. Akinetic-rigid and tremor-dominant Parkinson’s disease patients show different patterns of intrinsic brain activity. Parkinsonism Relat Disord [Internet]. 2015 [cited 2015 Jan 13];21:23–30. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25465747 | :::# Zhang J, Wei L, Hu X, Xie B, Zhang Y, Wu G-R, et al. Akinetic-rigid and tremor-dominant Parkinson’s disease patients show different patterns of intrinsic brain activity. Parkinsonism Relat Disord [Internet]. 2015 [cited 2015 Jan 13];21:23–30. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25465747 | ||
Revision as of 09:57, 28 April 2020
Ingram 2020 MitoFit Preprint Arch
| Ingram TL, Shephard F, Sarmad S, Ortori CA, Barrett DA and Chakrabart L (2020) Sex differences characterise inflammatory profiles of cerebellar mitochondria and are attenuated in Parkinson’s disease. MitoFit Preprint Arch doi:10.26124/mitofit:200002. |
»
Thomas L Ingram, Freya Shephard, Sarir Sarmad, Catherine A. Ortori, David A. Barrett and Lisa Chakrabarti. (2020) MitoFit Preprint Arch
Abstract: Version 1 (v1) 2020-04-23 doi:10.26124/mitofit:200002
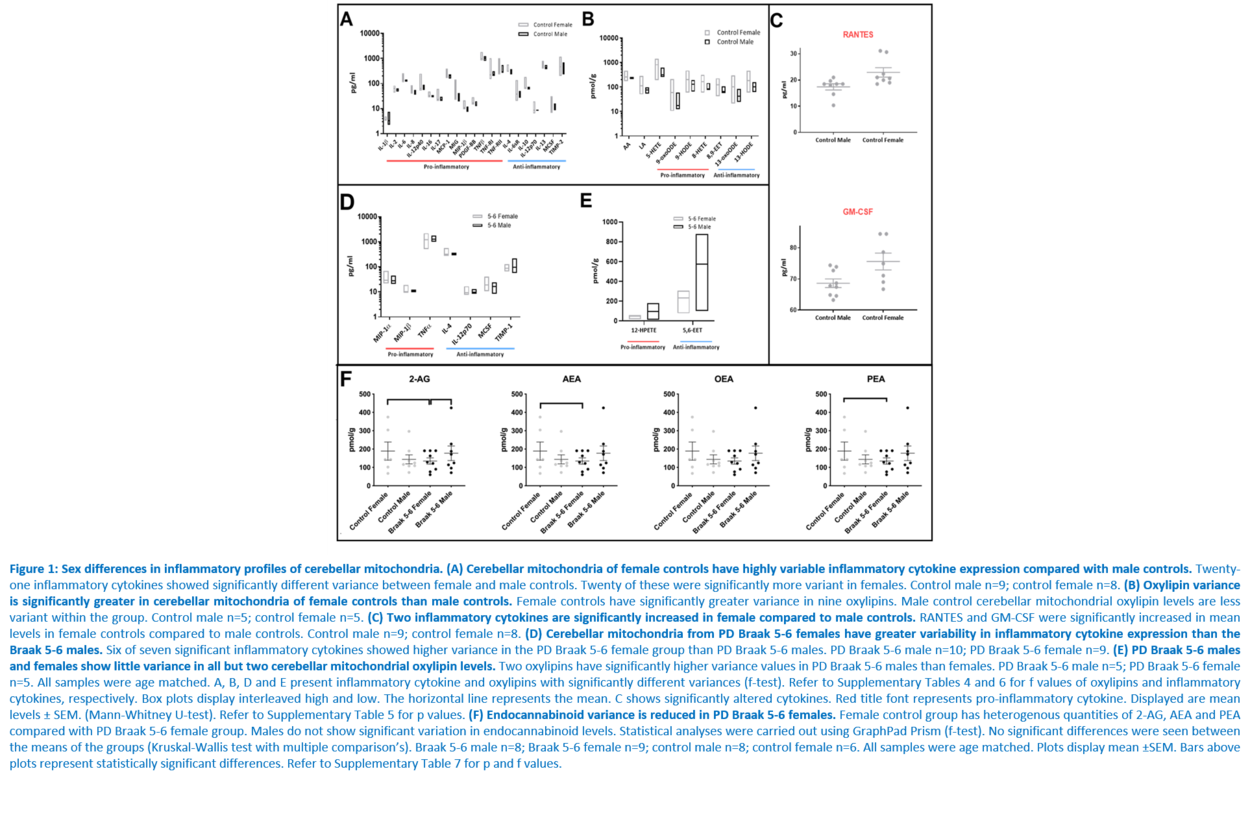
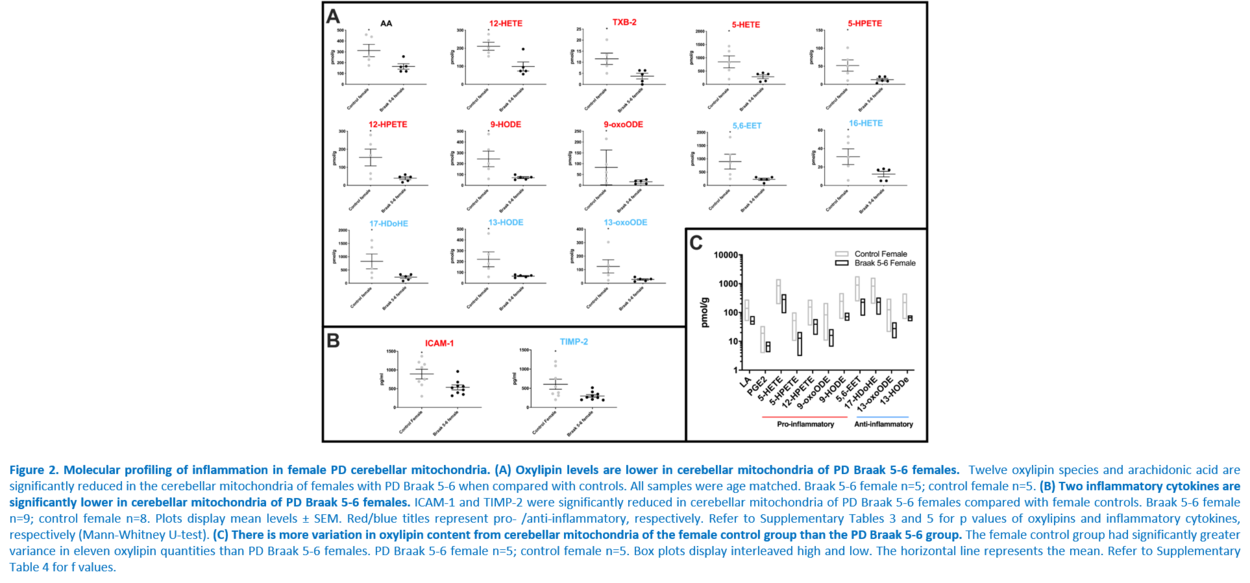
Response to inflammation is a key determinant in many diseases and their outcomes. Diseases that commonly affect older people are frequently associated with altered inflammatory processes. Neuroinflammation has been described in Parkinson’s disease (PD) brain and presents a potential therapeutic target. PD is characterised by the loss of dopaminergic neurons in the substantia nigra pars compacta and at the sub-cellular level, mitochondrial dysfunction is a key feature. However, there is evidence that a different region of the brain, the cerebellum, is involved in the pathophysiology of PD. We report relative levels of 40 pro- and anti-inflammatory cytokines measured in PD and control cerebellar mitochondria. These data were obtained by screening cytokine antibody arrays. In parallel, we present concentrations of 29 oxylipins and 4 endocannabinoids measured in mitochondrial fractions isolated from post-mortem PD cerebellum with age and sex matched controls. Our oxylipin and endocannabinoid data were acquired via quantitation by LC-ESI--MS/MS. The separate sample sets both show there are clearly different inflammatory profiles between the sexes in control samples. Sex specific profiles were not maintained in cerebellar mitochondria isolated from PD brains. One interpretation of our findings is that normally females have a wide-ranging inflammatory profile that can respond to or absorb the effects of increased levels of cytokines and oxylipins. These observations may have implications for other inflammatory diseases where the sexes are affected unequally in number or severity. • Keywords: Inflammation, Parkinson's disease, cerebellar mitochondria, sex differences • Bioblast editor: Iglesias-Gonzalez J
Figure
References
- Zhang J, Wei L, Hu X, Xie B, Zhang Y, Wu G-R, et al. Akinetic-rigid and tremor-dominant Parkinson’s disease patients show different patterns of intrinsic brain activity. Parkinsonism Relat Disord [Internet]. 2015 [cited 2015 Jan 13];21:23–30. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25465747
- Palmer SJ, Li J, Wang ZJ, McKeown MJ. Joint amplitude and connectivity compensatory mechanisms in Parkinson’s disease. Neuroscience [Internet]. 2010 [cited 2017 Oct 20];166:1110–8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20074617
- Dirkx MF, Zach H, van Nuland A, Bloem BR, Toni I, Helmich RC. Cerebral differences between dopamine-resistant and dopamine-responsive Parkinson’s tremor. Brain [Internet]. 2019 [cited 2019 Oct 28];142:3144–57. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31509182
- Seidel K, Bouzrou M, Heidemann N, Krüger R, Schöls L, den Dunnen WFA, et al. Involvement of the cerebellum in Parkinson disease and dementia with Lewy bodies. Ann Neurol. John Wiley and Sons Inc.; 2017;81:898–903.
- Wu T, Hallett M. The cerebellum in Parkinson’s disease. Brain [Internet]. 2013 [cited 2014 Jul 9];136:696–709. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23404337
- Stöger R, Scaife PJ, Shephard F, Chakrabarti L. Elevated 5hmC levels characterize DNA of the cerebellum in Parkinson’s disease. NPJ Park Dis [Internet]. 2017 [cited 2017 Oct 20];3:6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28649606
- Yoo HS, Choi YH, Chung SJ, Lee YH, Ye BS, Sohn YH, et al. Cerebellar connectivity in Parkinson’s disease with levodopa‐induced dyskinesia. Ann Clin Transl Neurol [Internet]. 2019 [cited 2019 Oct 28];acn3.50918. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31643140
- Kauppila TES, Kauppila JHK, Larsson NG. Mammalian Mitochondria and Aging: An Update. Cell Metab. Cell Press; 2017. p. 57–71.
- Area-Gomez E, Guardia-Laguarta C, Schon EA, Przedborski S. Mitochondria, OxPhos, and neurodegeneration: Cells are not just running out of gas. J. Clin. Invest. American Society for Clinical Investigation; 2019.
- Bose A, Beal MF. Mitochondrial dysfunction in Parkinson’s disease. J Neurochem [Internet]. 2016 [cited 2019 Sep 18];139:216–31. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27546335
- Sliter DA, Martinez J, Hao L, Chen X, Sun N, Fischer TD, et al. Parkin and PINK1 mitigate STING-induced inflammation. Nature. 2018.
- McWilliams TG, Prescott AR, Montava-Garriga L, Ball G, Singh F, Barini E, et al. Basal Mitophagy Occurs Independently of PINK1 in Mouse Tissues of High Metabolic Demand. Cell Metab. Cell Press; 2018;27:439-449.e5.
- . Hazeldine J, Dinsdale RJ, Harrison P, Lord JM. Traumatic Injury and Exposure to Mitochondrial-Derived Damage Associated Molecular Patterns Suppresses Neutrophil Extracellular Trap Formation. Front Immunol [Internet]. 2019 [cited 2019 Nov 11];10:685. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31001279
- Rieusset J. Mitochondria-associated membranes (MAMs): An emerging platform connecting energy and immune sensing to metabolic flexibility. Biochem Biophys Res Commun [Internet]. 2018 [cited 2019 Nov 11];500:35–44. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28647358
- Smith KM, Dahodwala N. Sex differences in Parkinson’s disease and other movement disorders. Exp Neurol [Internet]. 2014 [cited 2014 Oct 3];259:44–56. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24681088
- Siani F, Greco R, Levandis G, Ghezzi C, Daviddi F, Demartini C, et al. Influence of Estrogen Modulation on Glia Activation in a Murine Model of Parkinson’s Disease. Front Neurosci [Internet]. 2017 [cited 2017 Oct 30];11:306. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28620274
- Kumagai T, Nagayama H, Ota T, Nishiyama Y, Mishina M, Ueda M. Sex differences in the pharmacokinetics of levodopa in elderly patients with Parkinson disease. Clin Neuropharmacol [Internet]. [cited 2015 Jan 26];37:173–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25384078
- Wong A, Sagar DR, Ortori CA, Kendall DA, Chapman V, Barrett DA. Simultaneous tissue profiling of eicosanoid and endocannabinoid lipid families in a rat model of osteoarthritis. J Lipid Res [Internet]. 2014 [cited 2016 May 24];55:1902–13. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4617365&tool=pmcentrez&rendertype=abstract
- Liakh I, Pakiet A, Sledzinski T, Mika A. Modern Methods of Sample Preparation for the Analysis of Oxylipins in Biological Samples. Molecules [Internet]. Multidisciplinary Digital Publishing Institute (MDPI); 2019 [cited 2019 Oct 28];24. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31027298
- Rajamani A, Borkowski K, Akre S, Fernandez A, Newman JW, Simon SI, et al. Oxylipins in triglyceride-rich lipoproteins of dyslipidemic subjects promote endothelial inflammation following a high fat meal. Sci Rep [Internet]. 2019 [cited 2019 Oct 28];9:8655. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31209255
- Ramsden CE, Ringel A, Majchrzak-Hong SF, Yang J, Blanchard H, Zamora D, et al. Dietary linoleic acid-induced alterations in pro- and anti-nociceptive lipid autacoids. Mol Pain [Internet]. 2016 [cited 2019 Oct 28];12:174480691663638. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27030719
- Caligiuri SPB, Parikh M, Stamenkovic A, Pierce GN, Aukema HM. Dietary modulation of oxylipins in cardiovascular disease and aging. Am J Physiol Circ Physiol [Internet]. 2017 [cited 2019 Oct 28];313:H903–18. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28801523
- Dexter DT, Carter CJ, Wells FR, Javoy-Agid F, Agid Y, Lees A, et al. Basal Lipid Peroxidation in Substantia Nigra Is Increased in Parkinson’s Disease. J Neurochem [Internet]. John Wiley & Sons, Ltd (10.1111); 1989 [cited 2019 Aug 12];52:381–9. Available from: http://doi.wiley.com/10.1111/j.1471-4159.1989.tb09133.x
- De Franceschi G, Fecchio C, Sharon R, Schapira AH V., Proukakis C, Bellotti V, et al. α-Synuclein structural features inhibit harmful polyunsaturated fatty acid oxidation, suggesting roles in neuroprotection. J Biol Chem [Internet]. 2017 [cited 2019 Aug 12];292:6927–37. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28232489
- Lee H-J, Bazinet RP, Rapoport SI, Bhattacharjee AK. Brain arachidonic acid cascade enzymes are upregulated in a rat model of unilateral Parkinson disease. Neurochem Res [Internet]. 2010 [cited 2015 Aug 20];35:613–9. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2836388&tool=pmcentrez&rendertype=abstract
- Vivekanantham S, Shah S, Dewji R, Dewji A, Khatri C, Ologunde R. Neuroinflammation in Parkinson’s disease: role in neurodegeneration and tissue repair. Int J Neurosci [Internet]. 2015 [cited 2019 Nov 4];125:717–25. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25364880
- Refolo V, Stefanova N. Neuroinflammation and Glial Phenotypic Changes in Alpha-Synucleinopathies. Front Cell Neurosci [Internet]. 2019 [cited 2019 Nov 11];13:263. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31263402
- Guzman-Martinez L, Maccioni RB, Andrade V, Navarrete LP, Pastor MG, Ramos-Escobar N. Neuroinflammation as a Common Feature of Neurodegenerative Disorders. Front Pharmacol [Internet]. 2019 [cited 2019 Nov 11];10:1008. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31572186
- Haque ME, Akther M, Jakaria M, Kim I, Azam S, Choi D. Targeting the microglial NLRP3 inflammasome and its role in Parkinson’s disease. Mov Disord [Internet]. 2019 [cited 2019 Nov 11];mds.27874. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31680318
- Leitner GR, Wenzel TJ, Marshall N, Gates EJ, Klegeris A. Targeting toll-like receptor 4 to modulate neuroinflammation in central nervous system disorders. Expert Opin Ther Targets [Internet]. 2019 [cited 2019 Nov 11];23:865–82. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31580163
- Mattson MP, Arumugam T V. Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States. Cell Metab. Cell Press; 2018. p. 1176–99.
- Newcombe EA, Camats-Perna J, Silva ML, Valmas N, Huat TJ, Medeiros R. Inflammation: The link between comorbidities, genetics, and Alzheimer’s disease 11 Medical and Health Sciences 1109 Neurosciences 11 Medical and Health Sciences 1107 Immunology. J. Neuroinflammation. BioMed Central Ltd.; 2018.
- Alafuzoff I, Ince PG, Arzberger T, Al-Sarraj S, Bell J, Bodi I, et al. Staging/typing of Lewy body related alpha-synuclein pathology: a study of the BrainNet Europe Consortium. Acta Neuropathol [Internet]. 2009 [cited 2015 Aug 13];117:635–52. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19330340
- Shephard F, Greville-Heygate O, Marsh O, Anderson S, Chakrabarti L. A mitochondrial location for haemoglobins-Dynamic distribution in ageing and Parkinson’s disease. Mitochondrion [Internet]. 2013 [cited 2014 Jan 14];14:64–72. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24333691
- Sigma-Aldrich. Technical bulletin: Bradford Reagent B6916. 2013;3–8.
- Shephard F, Greville-Heygate O, Marsh O, Anderson S, Chakrabarti L. A mitochondrial location for haemoglobins--dynamic distribution in ageing and Parkinson’s disease. Mitochondrion [Internet]. 2014 [cited 2017 Oct 13];14:64–72. Available from: http://linkinghub.elsevier.com/retrieve/pii/S1567724913002778
- Richardson D, Ortori CA, Chapman V, Kendall DA, Barrett DA. Quantitative profiling of endocannabinoids and related compounds in rat brain using liquid chromatography-tandem electrospray ionization mass spectrometry. Anal Biochem [Internet]. 2007 [cited 2016 May 24];360:216–26. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17141174
- Mounsey RB, Mustafa S, Robinson L, Ross RA, Riedel G, Pertwee RG, et al. Increasing levels of the endocannabinoid 2-AG is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson’s disease. Exp Neurol [Internet]. 2015 [cited 2019 Feb 4];273:36–44. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26244281
- Balgoma D, Checa A, Sar DG, Snowden S, Wheelock CE. Quantitative metabolic profiling of lipid mediators. Mol Nutr Food Res [Internet]. 2013 [cited 2019 Oct 28];57:1359–77. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23828856
- Pochard C, Leclair-Visonneau L, Coron E, Neunlist M, Rolli-Derkinderen M, Derkinderen P. Cyclooxygenase 2 is upregulated in the gastrointestinal tract in Parkinson’s disease. Mov Disord [Internet]. John Wiley and Sons Inc.; 2018 [cited 2020 Apr 2];33:493–4. Available from: http://doi.wiley.com/10.1002/mds.27237
- Blesa J, Trigo-Damas I, Dileone M, del Rey NLG, Hernandez LF, Obeso JA. Compensatory mechanisms in Parkinson’s disease: Circuits adaptations and role in disease modification. Exp. Neurol. Academic Press Inc.; 2017. p. 148–61.
- Mirdamadi JL. Cerebellar Role in Parkinson’s Disease. J Neurophysiol [Internet]. 2016 [cited 2016 Jan 25];jn.01132.2015. Available from: http://jn.physiology.org/content/early/2016/01/15/jn.01132.2015.abstract
- Stöger R, Scaife PJ, Shephard F, Chakrabarti L. Elevated 5hmC levels characterize DNA of the cerebellum in Parkinson’s disease. npj Park Dis [Internet]. Nature Publishing Group; 2017 [cited 2017 Feb 21];3:6. Available from: http://www.nature.com/articles/s41531-017-0007-3
- Kaut O, Kuchelmeister K, Moehl C, Wüllner U. 5-methylcytosine and 5-hydroxymethylcytosine in brains of patients with multiple system atrophy and patients with Parkinson’s disease. J Chem Neuroanat. Elsevier B.V.; 2019;96:41–8.
- Sliter DA, Martinez J, Hao L, Chen X, Sun N, Fischer TD, et al. Parkin and PINK1 mitigate STING-induced inflammation. Nature [Internet]. 2018 [cited 2018 Aug 24]; Available from: http://www.ncbi.nlm.nih.gov/pubmed/30135585
- Newman LE, Shadel GS. Pink1/Parkin link inflammation, mitochondrial stress, and neurodegeneration. J. Cell Biol. NLM (Medline); 2018. p. 3327–9.
- Stojkovska I, Wagner BM, Morrison BE. Parkinson’s disease and enhanced inflammatory response. Exp. Biol. Med. SAGE Publications Inc.; 2015. p. 1387–95.
- Krashia P, Cordella A, Nobili A, La Barbera L, Federici M, Leuti A, et al. Blunting neuroinflammation with resolvin D1 prevents early pathology in a rat model of Parkinson’s disease. Nat Commun. Nature Publishing Group; 2019;10.
- Picillo M, Nicoletti A, Fetoni V, Garavaglia B, Barone P, Pellecchia MT. The relevance of gender in Parkinson’s disease: a review. J Neurol [Internet]. 2017 [cited 2017 Oct 30];264:1583–607. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28054129
- Weiduschat N, Mao X, Beal MF, Nirenberg MJ, Shungu DC, Henchcliffe C. Sex differences in cerebral energy metabolism in Parkinson’s disease: A phosphorus magnetic resonance spectroscopic imaging study. Parkinsonism Relat Disord [Internet]. 2014 [cited 2014 May 7]; Available from: http://www.sciencedirect.com/science/article/pii/S135380201400056X
- Caranci G, Piscopo P, Rivabene R, Traficante A, Riozzi B, Castellano AE, et al. Gender differences in Parkinson’s disease: focus on plasma α-synuclein. J Neural Transm [Internet]. 2013 [cited 2014 Sep 8];120:1209–15. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23328951
- McCarthy MM, Nugent BM, Lenz KM. Neuroimmunology and neuroepigenetics in the establishment of sex differences in the brain. Nat. Rev. Neurosci. Nature Publishing Group; 2017. p. 471–84.
- Goodfellow PN, Lovell-Badge R. SRY and Sex Determination in Mammals. Annu Rev Genet. Annual Reviews; 1993;27:71–92.
- Dewing P, Chiang CWK, Sinchak K, Sim H, Fernagut PO, Kelly S, et al. Direct regulation of adult brain function by the male-specific factor SRY. Curr Biol. Curr Biol; 2006;16:415–20.
- Lee J, Pinares-Garcia P, Loke H, Ham S, Vilain E, Harley VR. Sex-specific neuroprotection by inhibition of the Y-chromosome gene, SRY, in experimental Parkinson’s disease. Proc Natl Acad Sci U S A. National Academy of Sciences; 2019;116:16577–82.
- Chen W, Yan X, Lv H, Liu Y, He Z, Luo X. Gender differences in prevalence of LRRK2-associated Parkinson disease:A meta-analysis of observational studies. Neurosci Lett [Internet]. 2019 [cited 2020 Jan 3];134609. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31698024
- Erickson MA, Liang WS, Fernandez EG, Bullock KM, Thysell JA, Banks WA. Genetics and sex influence peripheral and central innate immune responses and blood-brain barrier integrity. Janigro D, editor. PLoS One [Internet]. 2018 [cited 2020 Jan 3];13:e0205769. Available from: http://dx.plos.org/10.1371/journal.pone.0205769
- Ferdouse A, Leng S, Winter T, Aukema HM. The Brain Oxylipin Profile Is Resistant to Modulation by Dietary n-6 and n-3 Polyunsaturated Fatty Acids in Male and Female Rats. Lipids. John Wiley and Sons Inc.; 2019;54:67–80.
- Pace S, Sautebin L, Werz O. Sex-biased eicosanoid biology: Impact for sex differences in inflammation and consequences for pharmacotherapy. Biochem. Pharmacol. Elsevier Inc.; 2017. p. 1–11.
- Giuliani N, Sansoni P, Girasole G, Vescovini R, Passeri G, Passeri M, et al. Serum interleukin-6, soluble interleukin-6 receptor and soluble gp130 exhibit different patterns of age- and menopause-related changes. Exp Gerontol. 2001;36:547–57.
- Scotland RS, Stables MJ, Madalli S, Watson P, Gilroy DW. Sex differences in resident immune cell phenotype underlie more efficient acute inflammatory responses in female mice. Blood. 2011;118:5918–27.
- Marriott I, Huet-Hudson YM. Sexual dimorphism in innate immune responses to infectious organisms. Immunol. Res. 2006. p. 177–92.
- Chandra G, Rangasamy SB, Roy A, Kordower JH, Pahan K. Neutralization of RANTES and Eotaxin Prevents the Loss of Dopaminergic Neurons in a Mouse Model of Parkinson Disease. J Biol Chem [Internet]. 2016 [cited 2019 Aug 9];291:15267–81. Available from: http://www.jbc.org/lookup/doi/10.1074/jbc.M116.714824
- Lucas SM, Rothwell NJ, Gibson RM. The role of inflammation in CNS injury and disease. Br. J. Pharmacol. Br J Pharmacol; 2006.
- Eikelenboom P, Van Exel E, Hoozemans JJM, Veerhuis R, Rozemuller AJM, Van Gool WA. Neuroinflammation - An early event in both the history and pathogenesis of Alzheimer’s disease. Neurodegener Dis. Neurodegener Dis; 2010. p. 38–41.
- Kang KH, Liou HH, Hour MJ, Liou HC, Fu WM. Protection of dopaminergic neurons by 5-lipoxygenase inhibitor. Neuropharmacology. Elsevier Ltd; 2013;73:380–7.
- Fischer R, Maier O, Siegemund M, Wajant H, Scheurich P, Pfizenmaier K. A TNF receptor 2 selective agonist rescues human neurons from oxidative stress-induced cell death. PLoS One. PLoS One; 2011;6.
- Garwood CJ, Cooper JD, Hanger DP, Noble W. Anti-inflammatory impact of minocycline in a mouse model of tauopathy. Front Psychiatry. Front Psychiatry; 2010;1.
Preprints for Gentle Science
» MitoFit Preprints - the Open Access preprint server for mitochondrial physiology and bioenergetics
Labels: MiParea: mt-Medicine
Pathology: Parkinson's
Organism: Human