Complex II ambiguities: Difference between revisions
No edit summary |
No edit summary |
||
| Line 5: | Line 5: | ||
}} | }} | ||
== FADH<sub>2</sub> and S-pathway == | == FADH<sub>2</sub> and S-pathway == | ||
[[File:Arnold, Finley 2022 CORRECTION.png|800px | :::: [[File:Arnold, Finley 2022 CORRECTION.png|800px|link=Arnold 2022 J Biol Chem]] | ||
:::: [[File:Martinez-Reyes, Chandel 2020 CORRECTION.png|800px|link=Martinez-Reyes 2020 Nat Commun]] | |||
:::: [[File:DeBerardinis, Chandel 2016 CORRECTION.png|800px|link=DeBerardinis 2016 Sci Adv]] | |||
:::: [[File:Sanchez et al 2001 CORRECTION.png|800px|link=Sanchez 2001 Br J Pharmacol]] | |||
:::: [[File:Himms-Hagen, Harper 2001 CORRECTION.png|250px|right|link=Himms-Hagen 2001 Exp Biol Med (Maywood)]] | |||
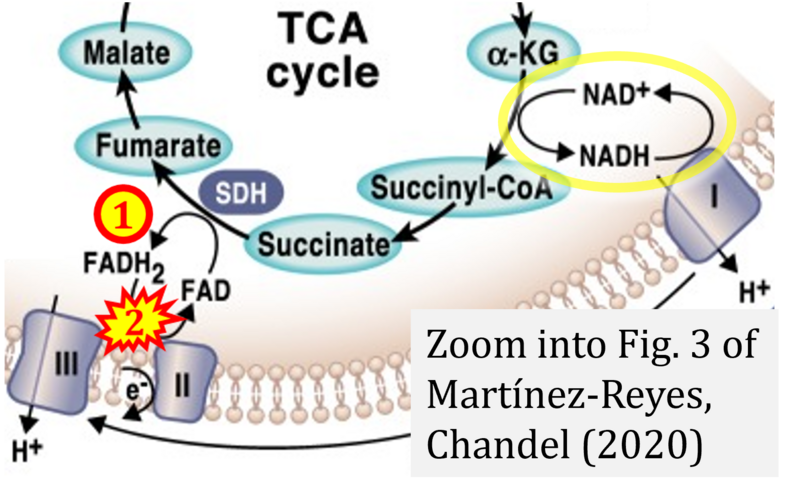
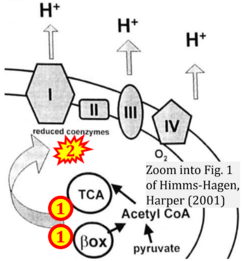
:::* FADH<sub>2</sub> appears in several publications as the substrate of CII in the electron transfer system. This commonly found error requires correction. For clarification, see page 48 in [[Gnaiger_2020_BEC_MitoPathways |Gnaiger (2020)]]. | :::* FADH<sub>2</sub> appears in several publications as the substrate of CII in the electron transfer system. This commonly found error requires correction. For clarification, see page 48 in [[Gnaiger_2020_BEC_MitoPathways |Gnaiger (2020)]]. | ||
::::* [[Arnold 2022 J Biol Chem]] | ::::* [[Arnold 2022 J Biol Chem]] | ||
| Line 11: | Line 15: | ||
::::* [[Yepez 2018 PLOS One]] | ::::* [[Yepez 2018 PLOS One]] | ||
::::* [[DeBerardinis 2016 Sci Adv]] | ::::* [[DeBerardinis 2016 Sci Adv]] | ||
::::* [[Himms-Hagen 2001 Exp Biol Med (Maywood)]] | |||
::::* [[Sanchez 2001 Br J Pharmacol]] | ::::* [[Sanchez 2001 Br J Pharmacol]] | ||
Revision as of 18:16, 16 February 2023
Description
The Succinate pathway is represented in the literature in some cases with a surprising confusion which warrants an analysis of Complex II ambiguities. The CII ambiguity has its roots in the narrative that reduced coenzymes (NADH and FADH2) feed electrons from the Krebs cycle into the membrane-bound electron transfer system. In corresponding ambiguous graphical representations, CII in the canonical ('forward') Krebs cycle is shown to reduce FAD to FADH2 (correct), yet CII in the membrane-bound electron transfer system is paradoxically represented as the site of oxidation of FADH2 to FAD. For clarification: "The substrate of CII is succinate, which is oxidized forming fumarate while reducing flavin adenine dinucleotide FAD to FADH2, with further electron transfer to the quinone pool. Whereas reduced NADH is a substrate of Complex I linked to dehydrogenases of the TCA cycle and mt-matrix upstream of CI, reduced FADH2 is a product of Complex II with downstream electron flow from CII to Q" (quote from Gnaiger 2020).
Abbreviation: CII ambiguities
Reference: Gnaiger E (2020) Mitochondrial pathways and respiratory control. An introduction to OXPHOS analysis. 5th ed. Bioenerg Commun 2020.2. https://doi.org/10.26124/bec:2020-0002
FADH2 and S-pathway
-
- FADH2 appears in several publications as the substrate of CII in the electron transfer system. This commonly found error requires correction. For clarification, see page 48 in Gnaiger (2020).
-
CII and fatty acid oxidation
- "Since mitochondrial Complex II also participates in the oxidation of fatty acids (6), .." (quote from [ Lemmi et al 1990]).
- Ref 6: Tzagoloff A (1982) Mitochondria. Plenum, New York.
- "Since mitochondrial Complex II also participates in the oxidation of fatty acids (6), .." (quote from [ Lemmi et al 1990]).
MitoPedia concepts: MiP concept
MitoPedia topics:
Enzyme,
Substrate and metabolite