Lin 2013 PLoS One
| Lin F, Chen Y, Levine R, Lee K, Yuan Y, Lin XN (2013) Improving fatty acid availability for bio-hydrocarbon production in Escherichia coli by metabolic engineering. PLoS One 8:e78595. https://doi.org/10.1371/journal.pone.0078595 |
Lin F, Chen Y, Levine R, Lee K, Yuan Y, Lin XN (2013) PLoS One
Abstract: Previous studies have demonstrated the feasibility of producing fatty-acid-derived hydrocarbons in Escherichia coli. However, product titers and yields remain low. In this work, we demonstrate new methods for improving fatty acid production by modifying central carbon metabolism and storing fatty acids in triacylglycerol. Based on suggestions from a computational model, we deleted seven genes involved in aerobic respiration, mixed-acid fermentation, and glyoxylate bypass (in the order of cyoA, nuoA, ndh, adhE, dld, pta, and iclR) to modify the central carbon metabolic/regulatory networks. These gene deletions led to increased total fatty acids, which were the highest in the mutants containing five or six gene knockouts. Additionally, when two key enzymes in the fatty acid biosynthesis pathway were over-expressed, we observed further increase in strain △cyoA△adhE△nuoA△ndh△pta△dld, leading to 202 mg/g dry cell weight of total fatty acids, ~250% of that in the wild-type strain. Meanwhile, we successfully introduced a triacylglycerol biosynthesis pathway into E. coli through heterologous expression of wax ester synthase/acyl-coenzyme:diacylglycerol acyltransferase (WS/DGAT) enzymes. The added pathway improved both the amount and fuel quality of the fatty acids. These new metabolic engineering strategies are providing promising directions for future investigation.
• Bioblast editor: Gnaiger E
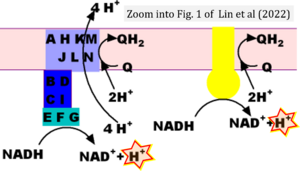
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.