Beutner 2014 PLoS One
| Beutner G, Eliseev RA, Porter GA Jr (2014) Initiation of electron transport chain activity in the embryonic heart coincides with the activation of mitochondrial complex 1 and the formation of supercomplexes. PLoS One 9:e113330. https://doi.org/10.1371/journal.pone.0113330 |
Beutner G, Eliseev RA, Porter GA Jr (2014) PLoS One
Abstract: Mitochondria provide energy in form of ATP in eukaryotic cells. However, it is not known when, during embryonic cardiac development, mitochondria become able to fulfill this function. To assess this, we measured mitochondrial oxygen consumption and the activity of the complexes (Cx) 1 and 2 of the electron transport chain (ETC) and used immunoprecipitation to follow the generation of mitochondrial supercomplexes. We show that in the heart of mouse embryos at embryonic day (E) 9.5, mitochondrial ETC activity and oxidative phosphorylation (OXPHOS) are not coupled, even though the complexes are present. We show that Cx-1 of the ETC is able to accept electrons from the Krebs cycle, but enzyme assays that specifically measure electron flow to ubiquinone or Cx-3 show no activity at this early embryonic stage. At E11.5, mitochondria appear functionally more mature; ETC activity and OXPHOS are coupled and respond to ETC inhibitors. In addition, the assembly of highly efficient respiratory supercomplexes containing Cx-1, -3, and -4, ubiquinone, and cytochrome c begins at E11.5, the exact time when Cx-1 becomes functional activated. At E13.5, ETC activity and OXPHOS of embryonic heart mitochondria are indistinguishable from adult mitochondria. In summary, our data suggest that between E9.5 and E11.5 dramatic changes occur in the mitochondria of the embryonic heart, which result in an increase in OXPHOS due to the activation of complex 1 and the formation of supercomplexes.
• Bioblast editor: Gnaiger E
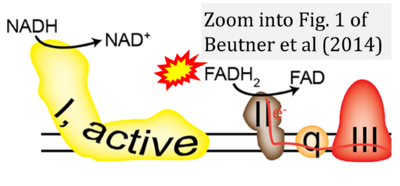
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Labels:
Enzyme: Complex II;succinate dehydrogenase